What is the IUPAC name of the following compound?
b. 4-ethyl-8-methyl-non-4-ene
c. 6-ethyl-2-methyl-non-5-ene
d. 7-methyl-3-propyl-oct-3-ene

b. 4-ethyl-8-methyl-non-4-ene
c. 6-ethyl-2-methyl-non-5-ene
d. 7-methyl-3-propyl-oct-3-ene

1 Answer
Aug 10, 2018
b. 4-ethyl-8-methyl-non-4-ene
Explanation:
The structure is an alkene, that means, the first thing we'll need to do is find the longest chain containing the double bond.
- That's the blue highlighted chain containing 9 carbon
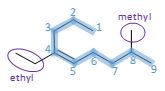
Then, we'll need to start counting from the end that is closest to the double bond.
- That's starting from the top rather than from the right. If we count following the labeled numbering, the double bond is between C4 and C5.
- However, if we count from the right, the double bond will be between C5 and C6. That's further away than C4-C5.
- So it's decided that it's non-4-ene.
Lastly, we'll place in the 2 substituents - ethyl at C4 and methyl at C8.
Putting everything together, the IUPAC name for this compound is 4-ethyl-8-methyl-non-4-ene .

