What is the mass number and the atomic number of Boron-11?
1 Answer
Explanation:
When dealing with an element's isotopes, it's important to remember that they are distinguished from ech other by the addition of the mass number to the name of said element.
In your case, the name boron-11 is used to designate an isotope of boron that has a mass number equal to
Since mass number is defined as the sum of the number of protons and the number of neutrons the atom has in its nucleus, it follows that you can write
#color(blue)(A = Z + "no. of neutrons")" "# , where
As you know, the identity of an element is given exclusively by the number of protons it has in its nucleus. This number represents the element's atomic number.
So, to identify the atomic number of boron, take quick look at a periodic table. Notice that boron,
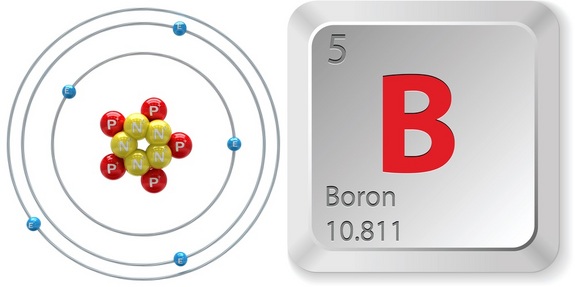 http://www.livescience.com/28674-boron.html
http://www.livescience.com/28674-boron.html
This means that any atom that has
So, the mass number of this boron isotope is equal to

