What is the mass number of a Calcium atom if it has 20 neutrons?
1 Answer
Explanation:
First thing first, you need to make sure that you have a clear understanding of what mass number actually is.
As you know, the nucleus of an atom contains protons, which are positively charged particles, and neutrons, which are neutral particles.
An atom's mass number will always tell you how many protons and neutrons its nucleus contains.
#color(blue)("mass number" = "no. of protons" + "no. of neutrons")#
In your case, you know that an atom of calcium,
This means that in order to get its mass number, all you need to know is how many protons are found in its nucleus.
As you know, the number of protons an atom contains in its nucleus is given by that atom's atomic number. A quick look in the periodic table will reveal that calcium has an atomic number equal to
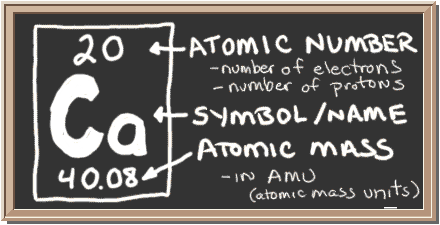
Now, in order for an atom to be an atom of calcium, it Must contain
Therefore, the mass number of this calcium isotope will be
#"mass number" = A = "20 protons" + "20 neutrons" = 40#
You are dealing with the calcium-40 isotope,

