What is the mechanism for the following reaction?
1 Answer
Here's what I get.
Explanation:
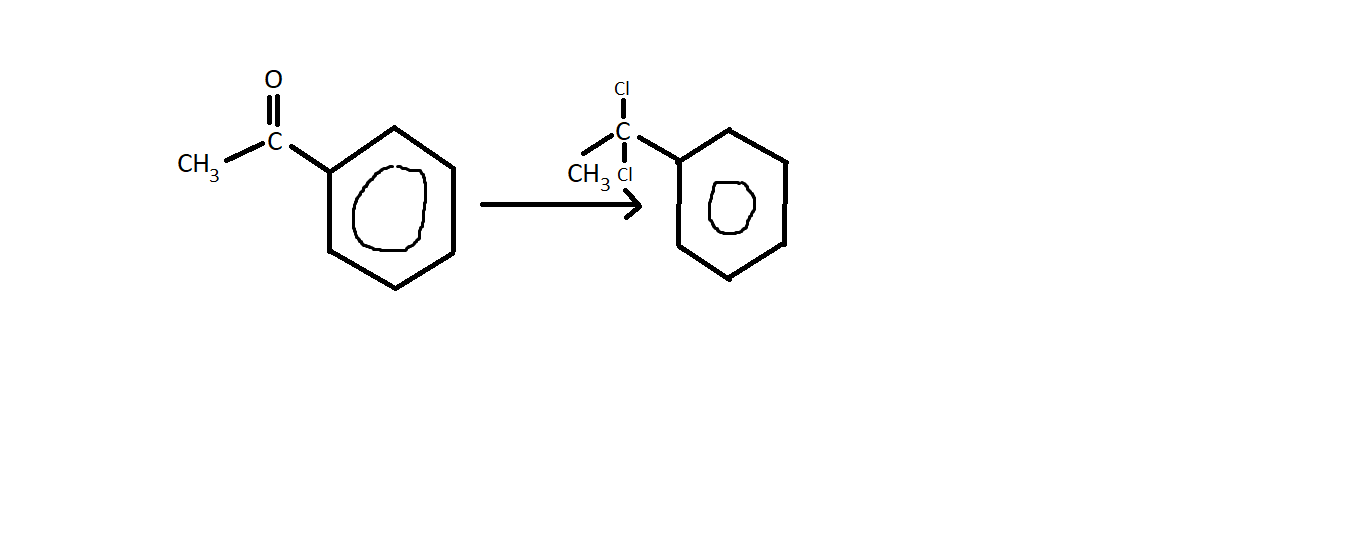
The overall reaction
A ketone reacts with
The reaction mechanism
Step 1
The
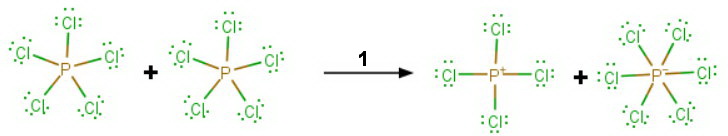
Step 2
The carbonyl oxygen attacks the
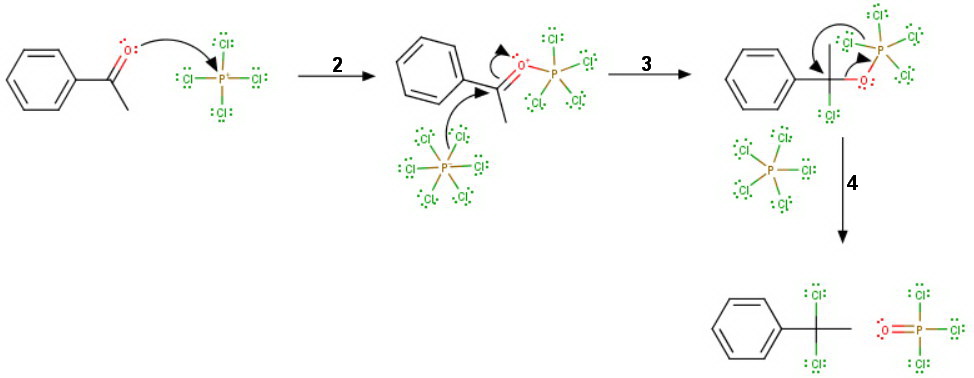
Step 3
A
Step 4
The new complex undergoes an intramolecular rearrangement to form the gem-dichloride.
An alternative to Step 4
Instead of being concerted, Step 4 could consist of three steps:
- Step 4a. A
#"Cl"^"-"# ion leaves the#"P"# atom of the complex. - Step 4b.
#"OPCl"_3# is an excellent leaving group. It departs in an#"S"_text(N)1# ionization to form a resonance-stabilized carbocation. - Step 4c. The
#"Cl"^"-"# ion attacks the carbocation and forms the gem-dichloride.

