What is the reaction for the formation of a hemiketal carbon in fructose? Also, is this the same as mutarotation?
1 Answer
Here's what I get.
Explanation:
Hemiketal formation
A hemiketal is a compound with the general formula
It is formed by the reaction of acetone with an alcohol.
(From Wikibooks)
In fructose, the

The product is called fructofuranose in analogy with the five-membered ring of furan.
The
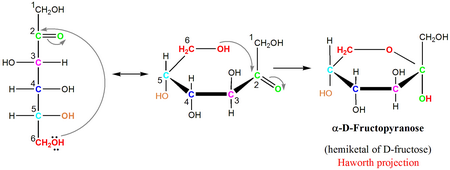
The product is called fructopyranose in analogy with the six-membered ring of pyran.
Mutarotation
The formation of a cyclic ketal generates a new stereocentre at C2.
Thus, both fructofuranose and fructopyranose exist in two stereoisomeric forms.
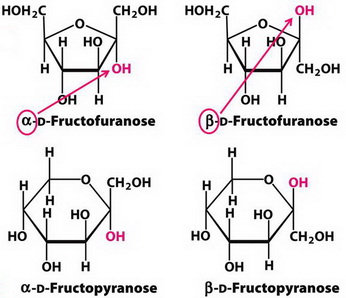
(Adapted from SlideShare)
In α- D-fructofuranose, the
The same is true of the pyranose forms.
The formation of the cyclic hemiketals is reversible.

Thus, if you start with a pure stereoisomer, in aqueous solution it will convert to an equilibrium mixture of all five forms.
Each component has its own specific rotation, so a solution of one stereoisomer will gradually change to that of the mixture.
This change in optical rotation as one isomer is converted to an equilibrium mixture is called mutarotation.
The major change is the ring size change for β-fructopyranose → β-fructofuranose, but the other isomers are present in small amounts.

