What is the strongest intermolecular force of attraction?
1 Answer
Quite probably
Explanation:
We speak of
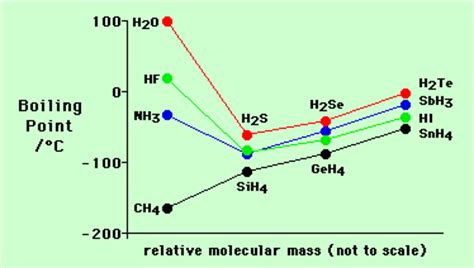
The boiling points of water, ammonia, and hydrogen fluoride, dwarf those of methane, and dwarf those of the heavier hydrides of the elements of Group 15, Group 16, and Group 17. And, CLEARLY, we may attribute this to the phenomenon of hydrogen-bonding, where hydrogen is bound to a strongly electronegative element, such as nitrogen, OR fluorine, OR oxygen. And the involatility of the water molecule, in which hydrogen bonding is MOST effective, is a clear consequence of this.
And so I maintain that the strongest intermolecular force of attraction is

