What is the volume of a container that holds 25.0 g of carbon gas at STP?
1 Answer
Jul 1, 2016
The container has a volume of
Explanation:
Since there are only one set of conditions and we are at STP, we must use the ideal gas law equation
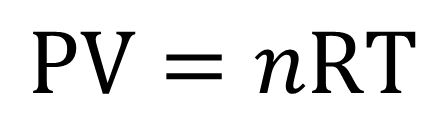
- P represents pressure (may have units of atm, depending on the units of the universal gas constnat)
- V represents volume (must have units of liters)
- n represents the number of moles
- R is the proportionality constant (has units of
#(Lxxatm)/ (molxxK)# ) - T represents the temperature, which must be in Kelvins.
Next, list your known and unknown variables. Our only unknown is the volume of
At STP, the temperature is 273K and the pressure is 1 atm. The proportionality constant, R, is equal to 0.0821
The only issue is the mass of
Now all we have to do is rearrange the equation and solve for V like so:

