What occurs as two atoms of fluorine combine to become a molecule of fluorine?
1 Answer
Mar 14, 2018
Explanation:
When two fluorine atoms combine, they become more stable, and form a molecule called
At room temperature and pressure, fluorine exists as a yellowish-greenish gas, and will almost react with anything just to get that extra electron.
Since the bonding of
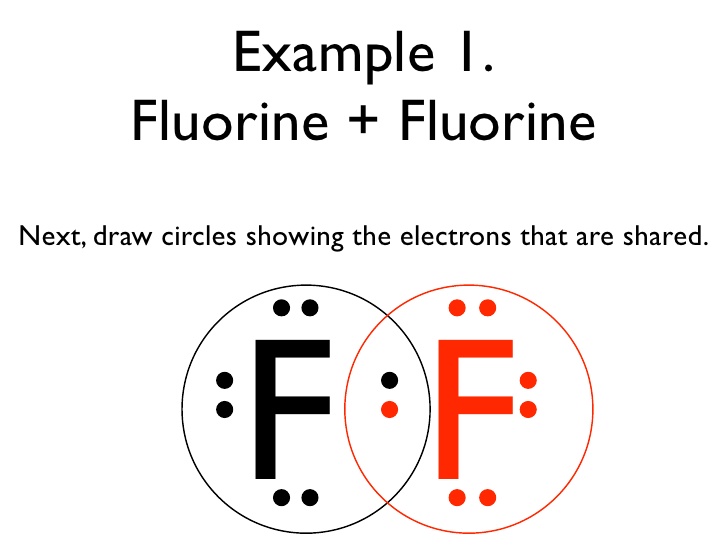
I hope this helps!

