What percentage does oxygen make up in the compound #MgSO_4#?
1 Answer
Oxygen makes up
Explanation:
In order to compute the mass percent of oxygen in
The formula mass of
The atomic mass of the oxygen atom is
Now, we have to use the following equation:
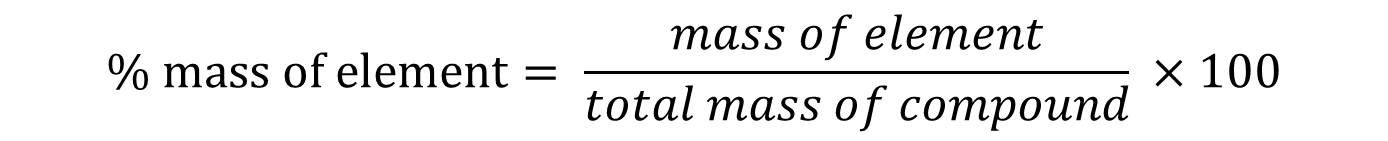
The numerator represents the mass of the desired atom, which is O in our case, and the denominator represents the mass of the entire compound. You just divide the two and multiply by 100 to obtain the percent composition:
I multiplied the atomic mass of oxygen by 4 because you have to account for all of the oxygen atoms in the formula.

