When 0.121 moles of HCl reacts with 0.399 moles of NH3, how much NH3 is consumed?
NH3+HCl -----> NH4Cl
NH3+HCl -----> NH4Cl
1 Answer
Oct 13, 2017
Approximately
Explanation:
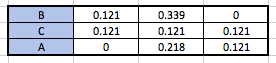
Fairly simple question. Consider the moles and coefficients of the reaction, and do some simple addition and subtraction to "get" the amount of

