When an acid is added to a basic solution, what happens to the pH of the solution?
1 Answer
May 1, 2017
Consider the graph of
Explanation:
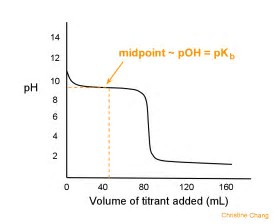
The graph depicts the titration of a strong base with a strong acid...
Well before equivalence, addition of the acid titrant only marginally affects solution

