When Uranium-238 decays, what does it decay into?
2 Answers
Below.
Explanation:
A nucleus of uranium-238 decays by alpha emission to form a daughter nucleus, thorium-234. This thorium, in turn, transforms into protactinium-234, and then undergoes beta-negative decay to produce uranium-234.
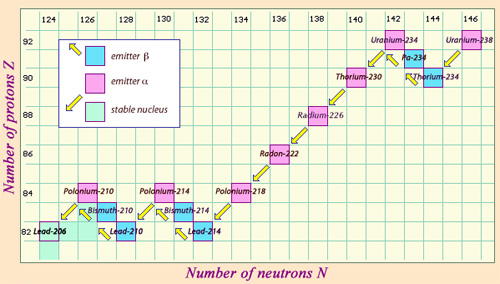 https://www.pinterest.de/pin/20618110767794606/
https://www.pinterest.de/pin/20618110767794606/
See Below
Explanation:
Big stuff like Uranium usually undergoes alpha particle decay. An alpha particle is a helium nucleus with 2 protons and 2 neutrons. So that means Uranium is going to lose two protons and 2 neutrons.
U-238 has 92 protons and (238-92 = 146 neutrons). If it loses 2 protons and 2 neutrons, it will then have 90 protons and 144 neutrons.
The element with 90 protons is Thorium, Th, and it will have a mass number of 144+90 = 234. So, you'll have Thorium-234 and an alpha particle running around.


