Which block of the periodic table is the largest?
1 Answer
The f-block is the largest block on the periodic table. They are the inner transition elements.
Explanation:
The f-block consists of two periods of 14 elements. There are 14 elements because the f-sublevels are being filled and an f-sublevel can contain a maximum of 14 electrons in its 7 orbitals. These elements are in periods 6 and 7 between lanthanium
The f-block is usually written below the main body of the periodic table because of its length, but some periodic tables, called extended or expanded periodic tables will place them within the body of the periodic table.
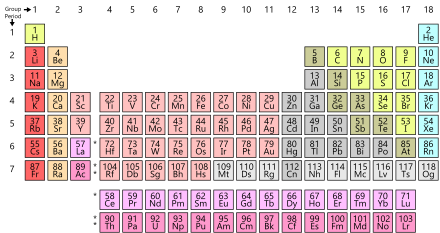 https://en.wikipedia.org/wiki/Periodic_table
https://en.wikipedia.org/wiki/Periodic_table
Extended Periodic Table
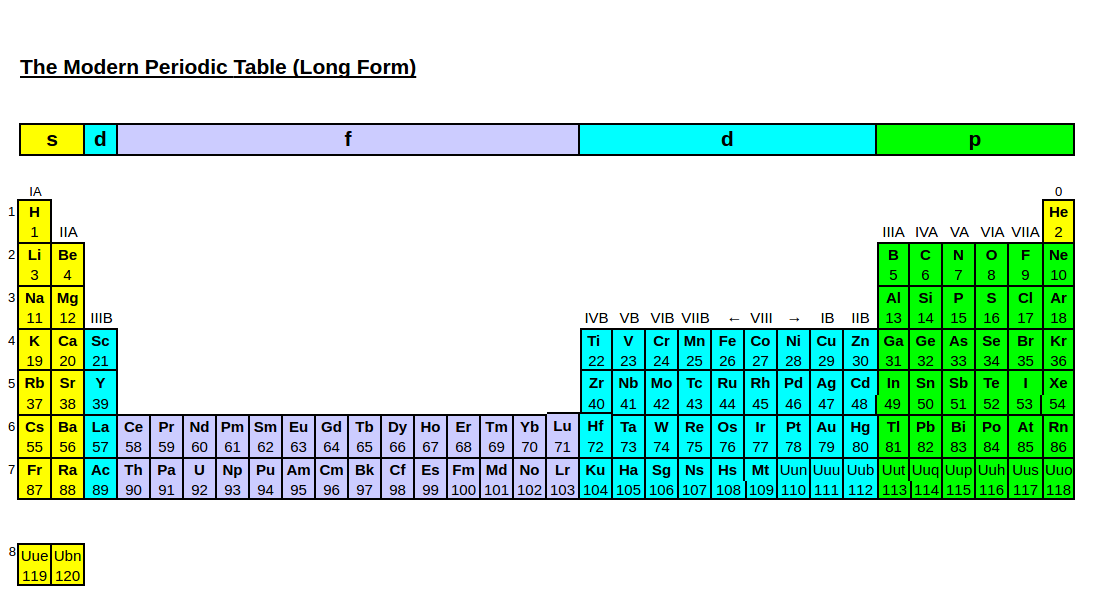 https://jeries.rihani.com/symmetry/index7.html
https://jeries.rihani.com/symmetry/index7.html

