Which element has the highest first ionization energy?
1 Answer
Ionization energies should increase across a period, and decrease down a group. Therefore the element with the highest 1st ionization should be helium.
Explanation:
Because we are physical scientists, while we can make predictions we should look at actual figures to check that we are not talking out of our posteriors. And look at this handy graph:
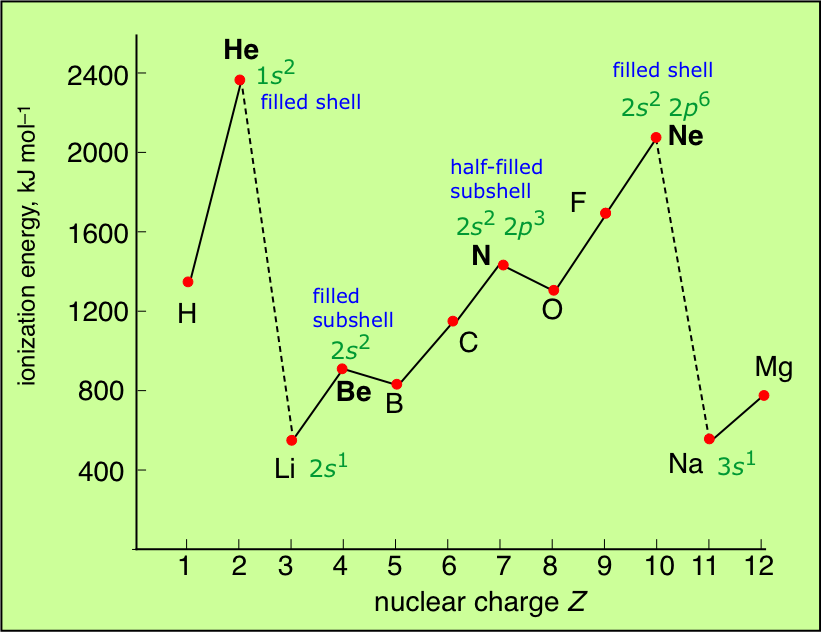
Is the graph consistent with the principles we discussed above?


