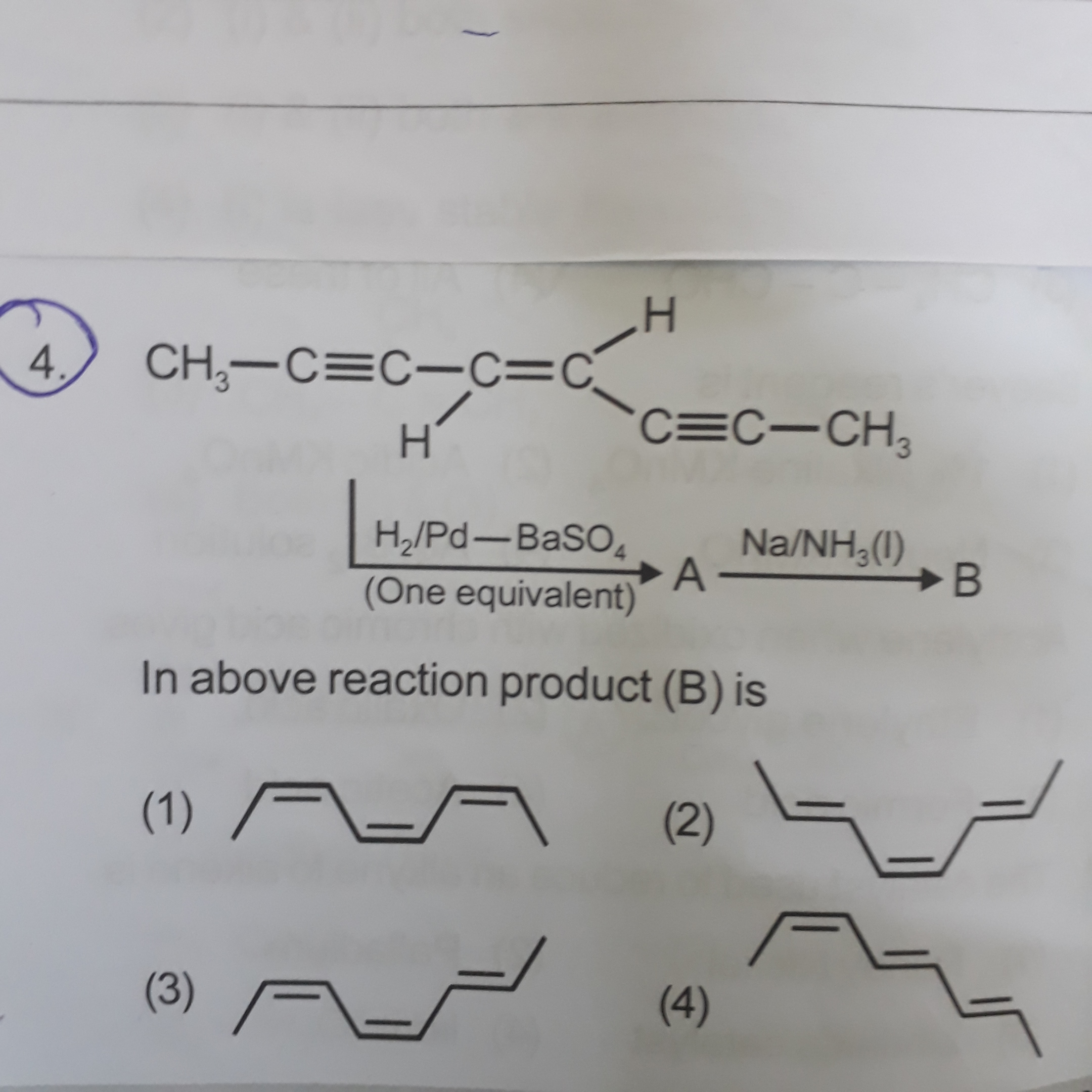
Which of the follwing is correct ? Please solve it
1 Answer
The answer is (4)
Explanation:
When one equivalent of Lindar's catalyst is given to the molecule, syn-addition takes place. We can choose anyone of the alkyne group as they are present symmetrically. Syn-Addition of hydrogen results in one of the alkyne group to get converted into cis-alkene.
Now, Birch reduction occurs at the other alkyne group, thus converting it into a trans alkene.
The alkene group present in the middle is already trans, so we have, the first alkyne converted into cis-alkene, the already present trans alkene group stays intact, and the last alkyne group becomes trans-alkene. The only option that fits our description is (4)

