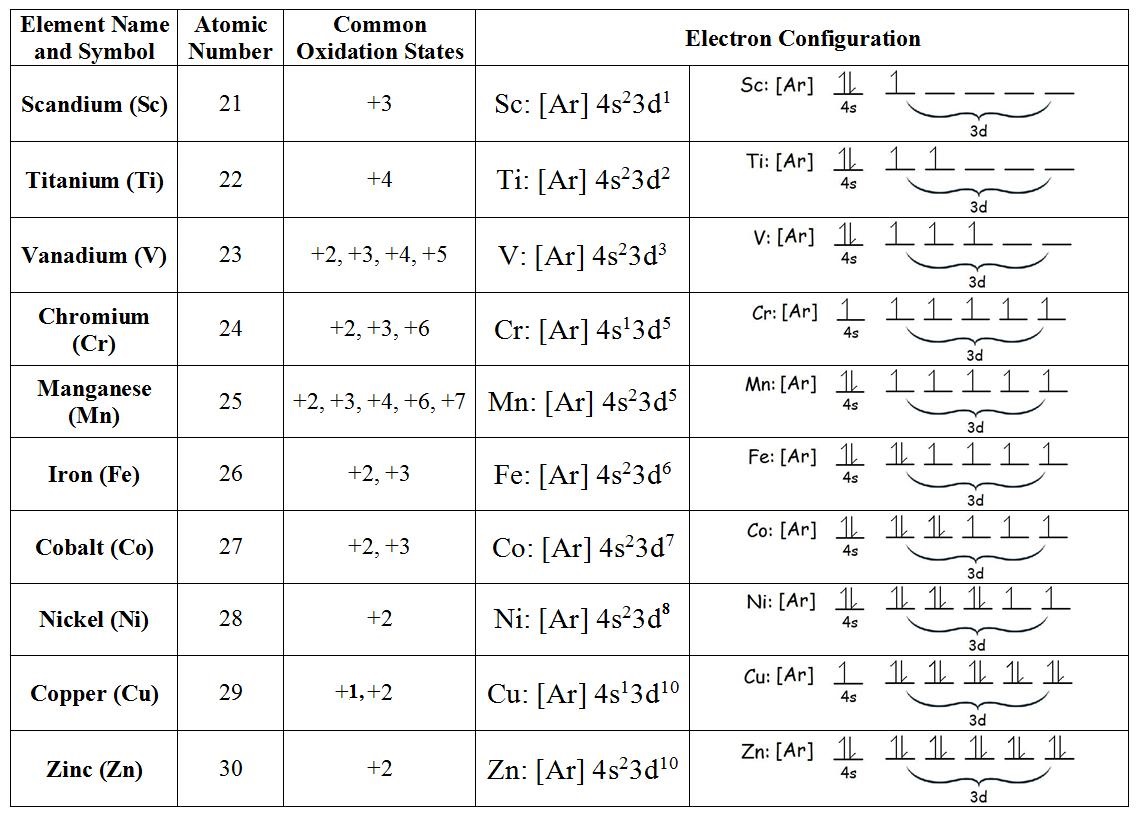
Which one of the elements with following outer electronic configurations may exhibit the largest number of oxidation states?
(1) #3d^5 4s^1#
(2) #3d^5 4s^2#
(3) #3d^2 4s^2#
(4) #3d^3 4s^2#
(1)
(2)
(3)
(4)
1 Answer
For transition metal elements, a general rule is => The max number of oxidation states = no. unpaired d-orbital electrons + the 2s electrons.
Explanation:
As noted, the maximum number of oxidation states a given transition metal is (generally) the number of unpaired d-orbitals plus the 2 s-orbital electrons. This then finds the transition elements with the higher numbers of oxidation states in the middle of the transition series.
For the question presented & written in order of Aufbau Diagram:
Example:
Iron has 4 unpaired electrons and 2 paired electrons. To find one of its oxidation states, we can use the formula:
Oxidation State of Fe = 4 + 2 = +6
Indeed, +6 is one of the oxidation states of iron, but it is very rare. Other possible oxidation states for iron includes: +5, +4, +3, and +2.
Here are some oxidation states of elements in the 1st Transition Series