Why are d and f block elements called transition elements?
1 Answer
They lie between the
They typically are considered to be the titanium family through the copper family.
Explanation:
Transition metals typically have a medium electronegativity, and as such a unique set of behaviours that vary from the
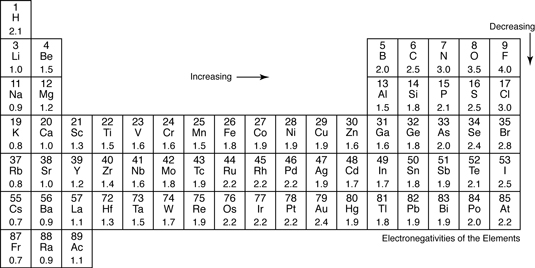
For example, gold and some of the other "noble metals" are highly resistant to corrosion and tend to remain in their elemental state rather than becoming oxidized, but the
[This is due to the high electronegativity values (gold is
Not all transition metals exhibit this property, but as electronegativity increases during the transition from metal to non-metal, the properties & behaviours begin to vary.
From an electronic point of view, the transition metal have valence electrons that include their


