Why are ionic compounds soluble in water?
1 Answer
Sep 22, 2014
You can look up here something about ionic bonding.
But, except that, here is a simple answer:
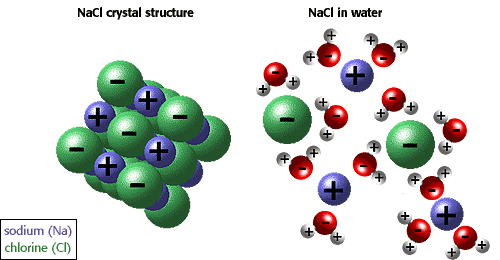 www.biology.arizona.edu
www.biology.arizona.edu
When ionic bonds form, one atom becomes positively charged (+), and the other one becomes negatively charged (-).
Water is a covalent polar compound (it has positive and negative poles). Also, ionic compound tend to form complex lattice networks and structures (see the picture).
When the salt is put in water, the water is pulling

