Why CaO is added in water body before pisciculture?
1 Answer
It neutralizes the water by increasing the pH of water(normally acidic)
Explanation:
{I suppose the topic should be biology.}
Liming helps prevent wide swings in pH. So, Liming may enhance the effect of fertilization. Liming also adds calcium and magnesium, which are important in animal physiology.
 https://www2.ca.uky.edu/wkrec/limingpondsaquaculture.htm
https://www2.ca.uky.edu/wkrec/limingpondsaquaculture.htm
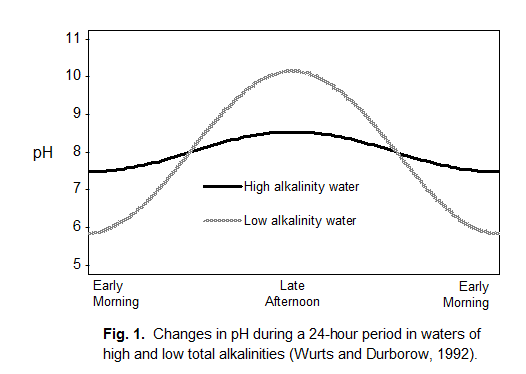
Fluctuations in pH are the result of the interplay of photosynthesis and respiration. Nighttime respiration increases
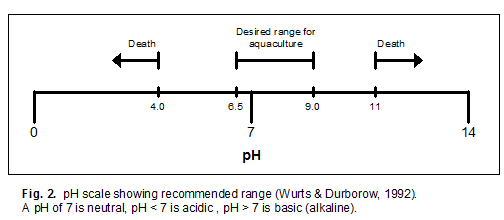
Large, daily changes in pH can stress aquatic animals . The desired alkalinity for many animals is 50 mg/L or higher. Liming to raise total alkalinity to the required or preferred ranges buffers the water and reduces swings in pH.
 https://www2.ca.uky.edu/wkrec/limingpondsaquaculture.htm
https://www2.ca.uky.edu/wkrec/limingpondsaquaculture.htm

