Why do all isotopes of an element have the same properties?
1 Answer
Sep 9, 2017
Actually, not all isotopes have the same properties.
Explanation:
Let me correct. Isotopes of an element has similar chemical properties but different physical properties.
Let's look at Hydrogen.
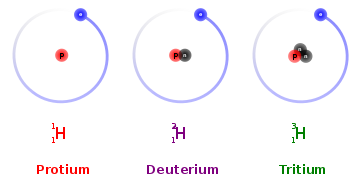
All 3 isotopes shown here have the same number of electrons, and hence it would have similar chemical properties. Since the isotopes have different number of neutrons, their masses would be different and hence different physical properties.

