Why do halogens do not add to double bond in allylic halogenation?
1 Answer
It's all a question of relative rates.
Explanation:
Bromine addition vs. allylic substitution
Consider the reaction
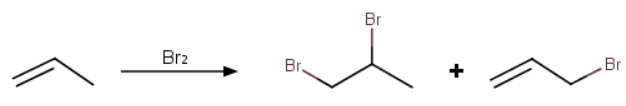
Why is the product almost exclusively the 1,2-dibromide?
Relative rates
The rate-determining step in an addition reaction is the attack of a bromine molecule to form a cyclic bromonium ion.
The rate-determining step in a substitution reaction is the attack of a bromine atom on an alkane.
At any one time, we have one equivalent of
If we could decrease the concentration of bromine molecules, the rate of addition would decrease, and the relative rate of substitution ould increase.
Allylic bromination
We can use
Then
Thus, the concentration of bromine stays low and allows the free-radical substitution to out-compete the alkene addition.

