Why do oil droplets lose an electron when they drop in Millikan's oil drop experiment?
1 Answer
Jun 22, 2018
They don't!
Explanation:
Millikan's graduate student Harvey Fetcher sprayed oil droplets into an apparatus and allowed them to fall through a pinhole into a chamber between two charged plates.
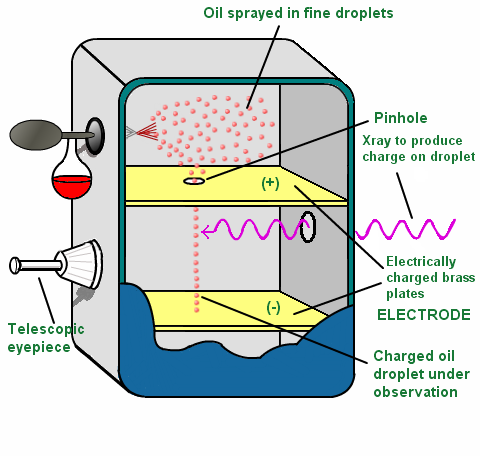
The droplets gained a negative charge in two ways:
- through friction with the nozzle
- by ionizing radiation from X-rays
The droplets fell because of gravity, not because they lost electrons.
Fletcher could adjust the voltage to make the upward attraction of the positive plate match the downward force of gravity.
He was then able to calculate the charge on the drop.
Here's a video that explains the oil-drop experiment.

