Why don't ionic compounds have electrical conductivity as a solid but they do as a liquid?
1 Answer
Solid ionic compounds do not have electrical conductivity because the ions are not free to move. In a liquid, the ionic compound dissociates into its respective ions.
Explanation:
Electrical conductivity requires the movement of charged particles. The common unit for charge is the Coulomb (
In the solid state, electrostatic forces hold the ions together in a crystal lattice structure, which is, in short, a 3D interconnected ion network.
A solid ionic compound does not conduct electricity because the ions are not free to move.
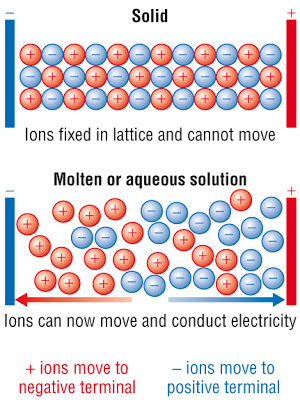
This flow of ions depicted immediately above is an electric current. You can tell because if we use
Therefore, charge-motion is required for there to be electric current.
When we heat an ionic compound to its melting point, the heat energy actually breaks the electrostatic forces between the ions, loosening the structure into something more like a liquid; the positive ions can move to the negative terminal, and the negative ions are free to move to the positive terminal, as depicted immediately above.
Since melting many ionic solids takes a lot of heat, we call this the molten state. Thus, ionic compounds can conduct electricity in the molten state.
They can also conduct electricity when dissolved in water; as they will dissociate into their ions, having the ability to conduct electricity (as they may move around freely, being electrolytes in solution).


