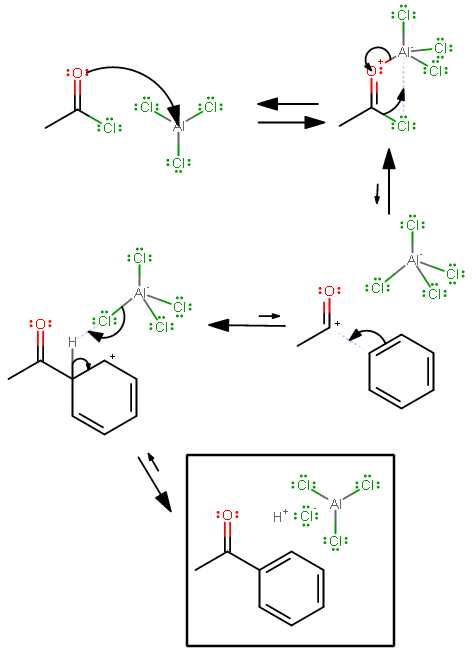
Due to the electronegativity differences between #"Cl"# (#3.16#) and #"Al"# (#1.61#), #"Cl"# makes a good electron-withdrawing group in #"AlCl"_3#.
Furthermore, #"Al"# must access both its #2s# and its three #2p# orbitals to bond, so it uses #sp^3# hybridization (one #2s# and three #2p# orbitals), giving it four bonding orbitals (one of which is empty as #"AlCl"_3#). This allows it to form a fourth bond and acquire a tetrahedral structure as #"AlCl"_4^(-)#.
With one empty orbital and three electron-withdrawing #"Cl"# atoms attached, the compound is thus an electron-acceptor at the #"Al"# center. By definition, that is a Lewis Acid.
Here is an example of Friedel-Crafts Acylation that shows the Lewis Acid behavior of #"Al"# in #"AlCl"_3#: