Why is the first ionization energy of a nonmetal significantly higher than that of an alkali metal?
1 Answer
Refer to the explanation.
Explanation:
The first ionization energy of an element is the energy needed to remove the outermost (valence) electron from a neutral atom in the gaseous state to form a cation.
The first ionization energy of an element has an inverse relationship to its atomic radius.
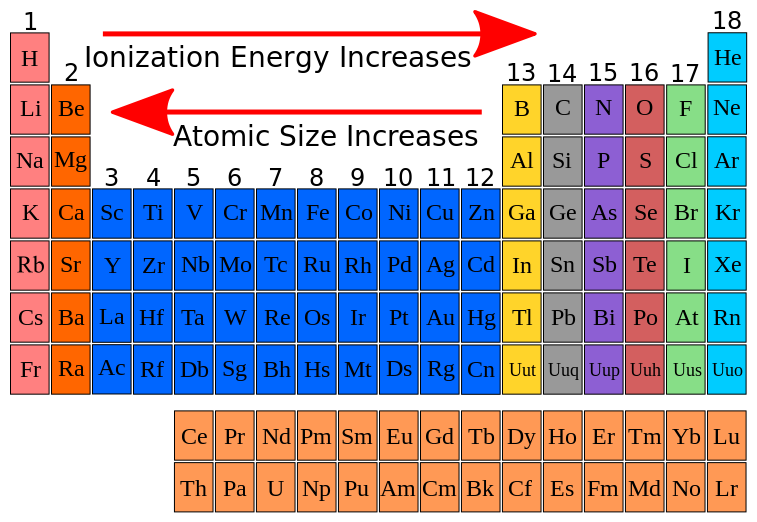
Across a period from left to right on the periodic table, first ionization energy increases and atomic radius decreases. Down a group, first ionization energy decreases and atomic radius increases.
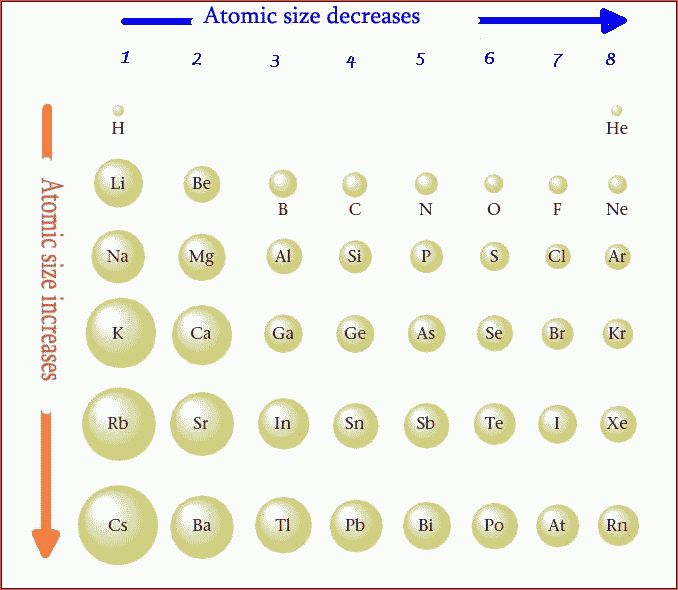
The atoms of the alkali metals have larger radii than those of nonmetals. Because of this, the attraction of the positively charged atomic nucleus for the valence electron is much less than that of a nonmetal.
Therefore, the first ionization energy of an alkali metal is much less than that of a nonmetal. Because of this, alkali metals lose their single valence electron which produces a cation with a charge of
The atoms of nonmetals have smaller radii, therefore the positively charged nucleus has a greater attraction for the valence electrons.
So the first ionization energy for a nonmetal is much greater than that of an alkali metal, so nonmetals do not lose electrons in an ionic bond, but instead gain one or more electrons and form anions. An example is a neutral chlorine (Cl) atom, also in period 3, which has an atomic radius of
Refer to the diagram below.
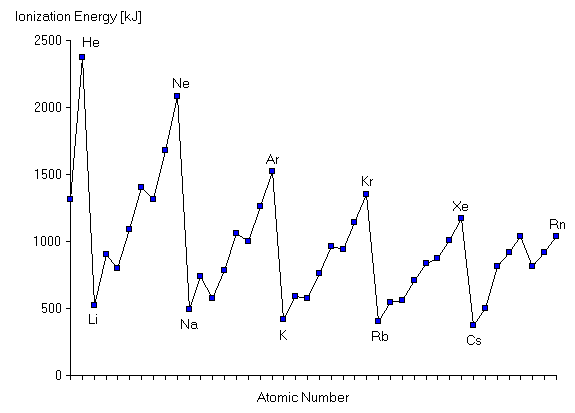
The first ionization energy increases across a period.


