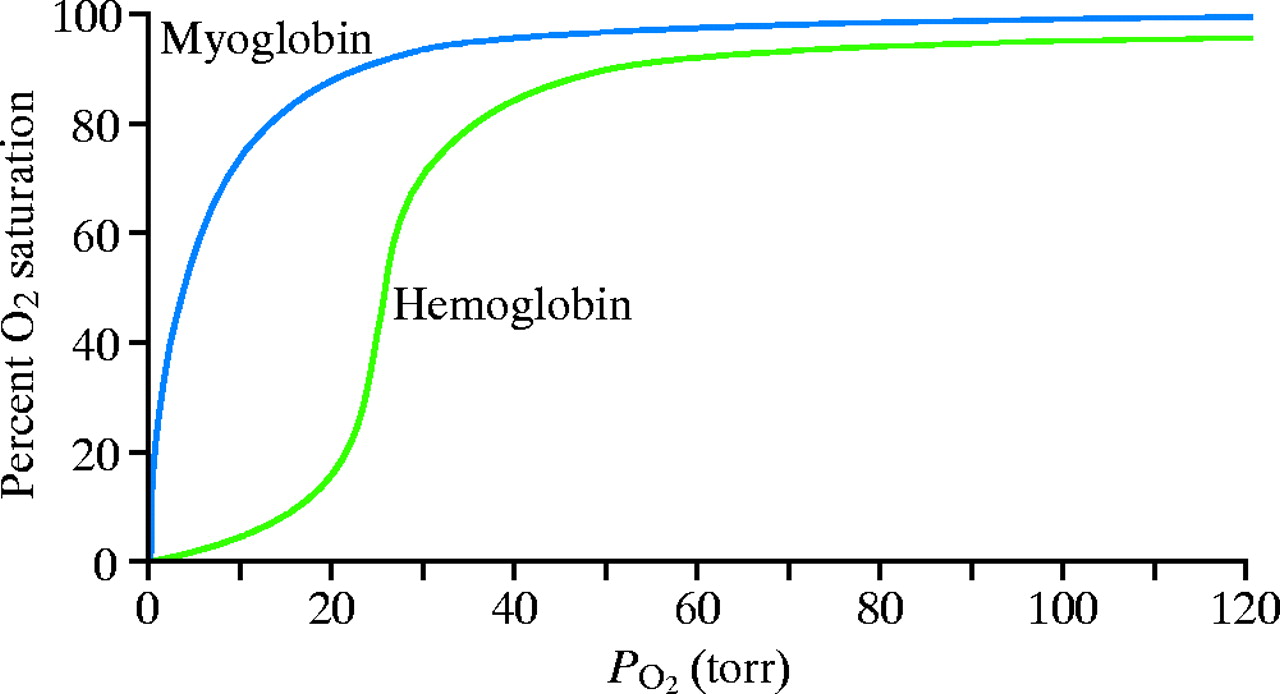
Why might an oxygen dissociation curve be sigmoidal?
1 Answer
Because it is associated with cooperative oxygen binding.
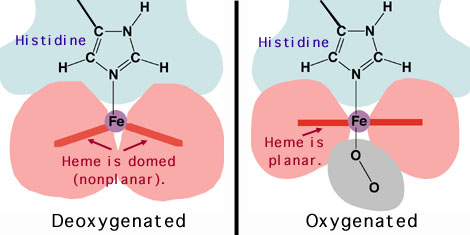
NONCOOPERATIVE VS. COOPERATIVE OXYGEN BINDING
Noncooperative oxygen binding is commonly associated with myoglobin. It is a monomer. It has a hyperbolic oxygen binding curve and does NOT have cooperative oxygen binding. This is described as:
#"Y"_(O_2) = ("P"_(O_2))/("K"_D + "P"_(O_2))# where
#"Y"# is the fractional saturation (y-axis),#"P"_(O_2)# is the partial pressure of oxygen in#"torr"# (x-axis), and#"K"_D# is the dissociation constant for binding events.#"K"_D# is smaller for higher binding affinities.
Cooperative oxygen binding is basically an effect where oxygen binding affinity can change depending on how much oxygen is bound, and this is described via a sigmoidal binding curve.
HEMOGLOBIN
Hemoglobin, an
#"Y"_(O_2) = ("P"_(O_2)^n)/("P"_50^n + "P"_(O_2)^n)# where
#"Y"# is the fractional saturation (y-axis),#"P"_(O_2)# is the partial pressure of oxygen in#"torr"# (x-axis),#"P"_50# is the partial pressure of oxygen when#"K"_D = "P"_(O_2)# , and#"K"_D# is the dissociation constant for binding events.#n <= 4# for Hemoglobin, and#"K"_D# is smaller for higher binding affinities.
And the binding curve looks like:
COOPERATIVE OXYGEN BINDING
Basically, cooperative oxygen binding means that at low quantities of oxygen, binding affinity is low and fractional saturation is also low, while at the same time, at high quantities of oxygen, binding affinity is high and fractional saturation is also high.
This is great because hemoglobin can bind oxygen well when there is a lot around, and release it well when there isn't a lot of oxygen around. This makes it easier for it to perform its job as an oxygen transport protein.
Non-mathematically, this can be described by noting that oxygen is a homotypic effector/regulator, so once it binds to the