You have 40g of sodium chloride. What is the minimum amount of water required to dissolve the salt at 30oC?
1 Answer
Jun 18, 2018
The minimum mass of water is 111 g.
Explanation:
Step 1. Determine the solubility of
I am guessing that "they" are asking you to get the information from a solubility curve for sodium chloride like the one below.
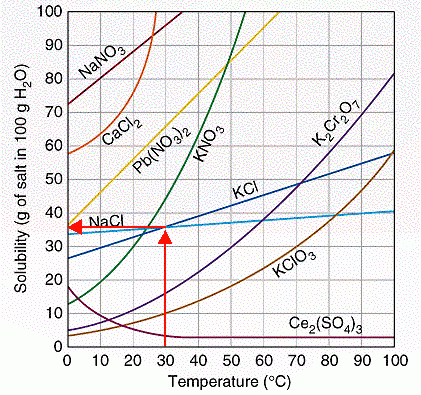
(Adapted from http://www.sciencegeek.net/Chemistry/taters/solubility)
From 30 °C on the horizontal axis, draw a vertical line up until it hits the solubility curve (blue) for
Then draw a horizontal line to the left until it meets the vertical axis, and read the solubility.
The solubility is
Step 2. Calculate the mass of water required

