1). What is the Lewis structure for SeF4? 2). What is its electron geometry? 3). What is its molecular geometry? 4. What is its hybridization? 5. How would you classify it in the AXE system? 6. What are the ideal bond angles?
1 Answer
Mar 22, 2014
1) See below; 2) trigonal bipyramidal; 3) see-saw; 4) sp³d; 5) AX₄E; 6) see below.
Explanation:
1) 1 Se + 4 Br = 6 + 4×7 = 34 valence electrons. Se must expand its octet. This gives Se four bond pairs and one lone pair. The Lewis structure is like that of SeF₄.
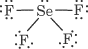
2). The electron arrangement for five electron pairs is trigonal bipyramidal.
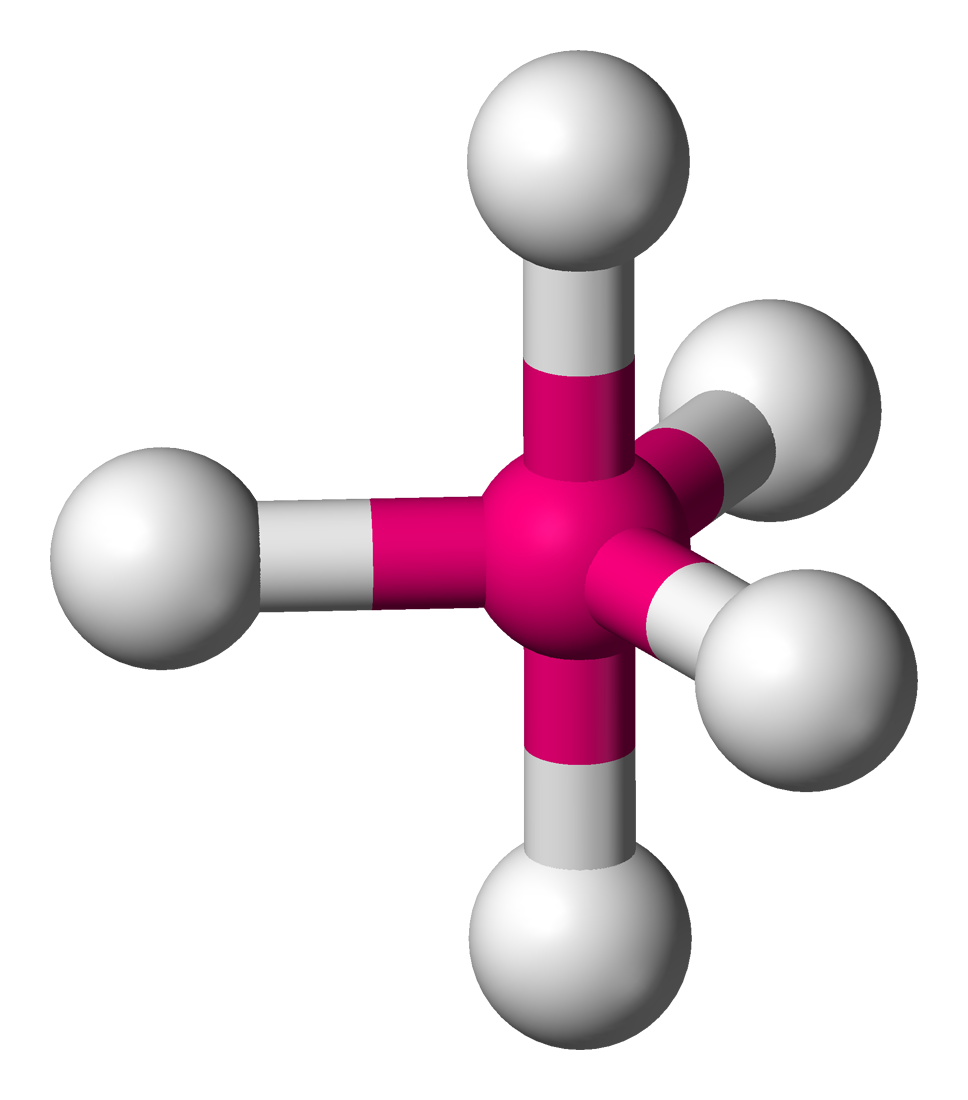
3) The bulky lone pair occupies an equatorial position.

The molecular shape considers only the Se-Br bonds. As with SF₄, the shape is a see-saw.
4) The hybridization that corresponds to five electron pairs is sp³d.
5) The VSEPR notation is AX₄E.
6) The bond angles are:
axial-axial = > 180°;
equatorial-equatorial = < 120°;
axial-equatorial = < 90°.

You can watch a video on trigonal bipyramidal geometry here:

