When the soft drink can is opened bubbles appear :can this be use to explain the principle behind effervescence when antacid tablets are added to water?
1 Answer
Quick answer: NO.
Explanation:
The bubbles that appear when a soft drink can is opened are the result of a physical change.
The carbon dioxide is dissolved in the contents of a soft drink can under pressure.
The concentration of carbon dioxide is given by Henry's Law,
where
When the container is opened, the pressure is released, the solubility decreases, and bubbles of the gas appear.
 Soft Drink
Soft Drink
(from physics.stackexchange.com)
The effervescence that occurs when antacid tablets are added to water is the result of a chemical change.
Effervescent tablets contain acid substances (e.g. citric acid) and carbonates or bicarbonates.
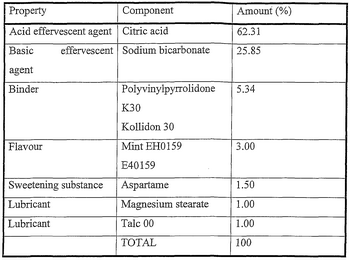 Effervescent
Effervescent
(from www.google.com)
They react rapidly in the presence of water by releasing carbon dioxide.
 Tablet
Tablet
(from www.losan-pharma.de)

