What volume of 12 M #HCl# solution is needed to prepare 5 liters of 0.0250 M solution?
1 Answer
Jul 10, 2016
Explanation:
In order to get the answer, you have to use the dilution equation:
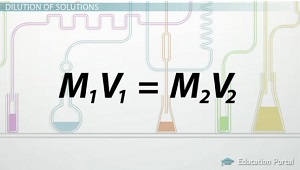
* TIP. * Whenever you are going from a highly concentrated substance to a less concentrated substance by increasing the volume of the solvent (dilution) you always use this equation.
We know

