The frequency of radiation emitted when electron falls from n=4 to n=1 in a hydrogen atom will be (given ionization energy of H = #2.18 xx 10^(-18) "J/atom"# and #h = 6.626 xx 10^(-34) "J"cdot"s"#)?
1 Answer
#nu_(4->1) = 3.084 xx 10^(15) "s"^(-1)#
Electronic transitions in the hydrogen atom are given by the Rydberg equation:
#bb(DeltaE = -R_H(1/n_f^2 - 1/n_i^2))# where:
#R_H = 2.18 xx 10^(-18) "J/H atom"# is the Rydberg constant. Yes, it is the ionization energy of hydrogen atom as well!#h = 6.626 xx 10^(-34) "J"cdot"s"# is Planck's constant.#n_k# is the principal quantum number for the#k# th state. Thus,#n_f# indicates the destination energy level, and#n_i# indicates the initial energy level.
So,
#DeltaE = -2.18 xx 10^(-18) "J/H atom" xx (1/1^2 - 1/4^2)# which should be negative with respect to the system! (why?)
...and we do indeed get a negative number...
#DeltaE = -2.044 xx 10^(-18) "J/H atom"#
The energy released from the
#|DeltaE| = E_"photon" = hnu# where
#nu# is the frequency in#"s"^(-1)# .
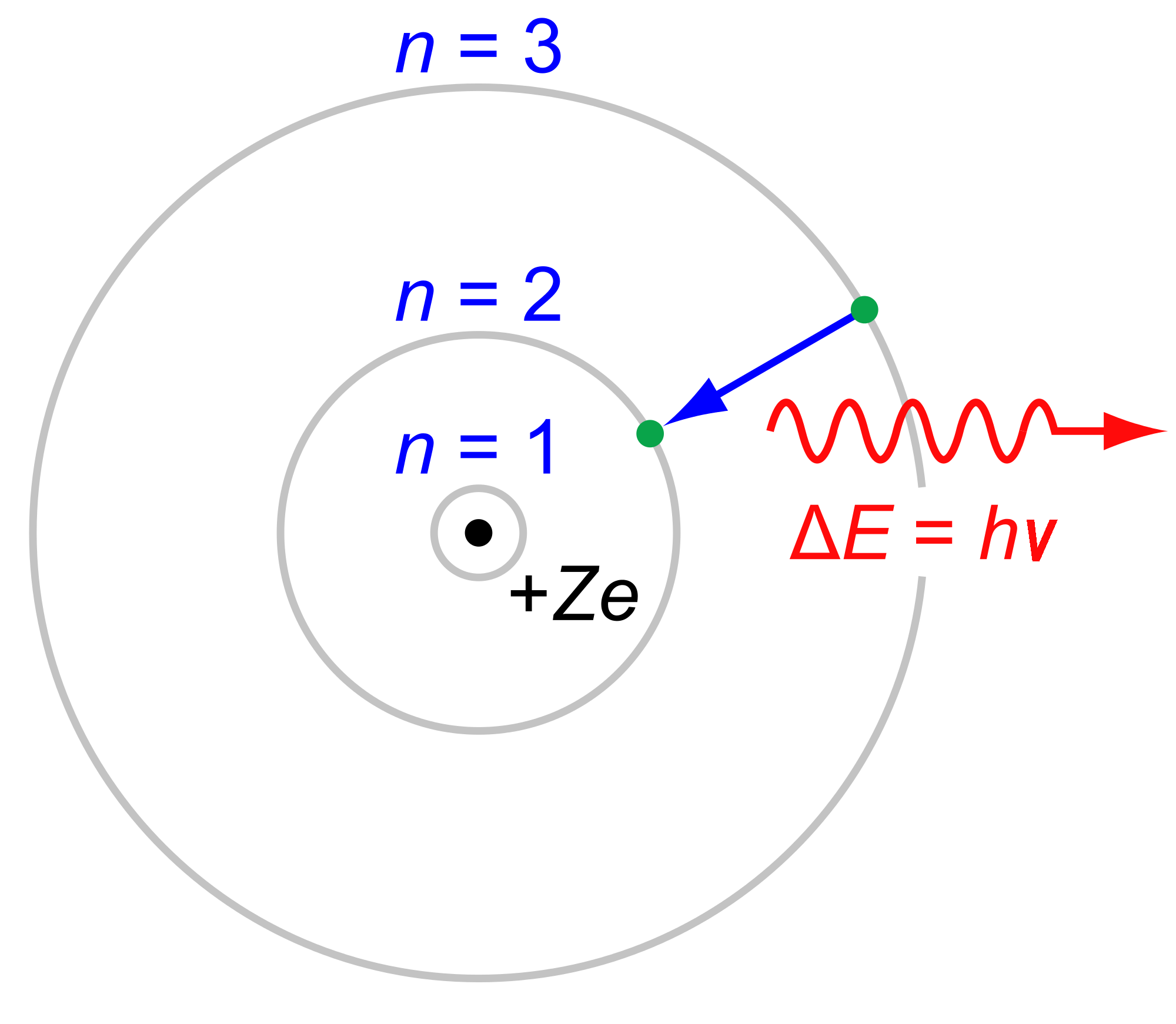
Thus, the energy per photon is:
#E_"photon" = +2.044 xx 10^(-18) "J/H atom"#
#= hnu#
and the frequency is therefore:
#color(blue)(nu) = E_"photon"/h = (+2.044 xx 10^(-18) cancel"J"/cancel"H atom" xx cancel"1 H atom"/"photon")/(6.626 xx 10^(-34) cancel"J"cdot"s")#
#= color(blue)ul(3.084 xx 10^(15) "s"^(-1))#

