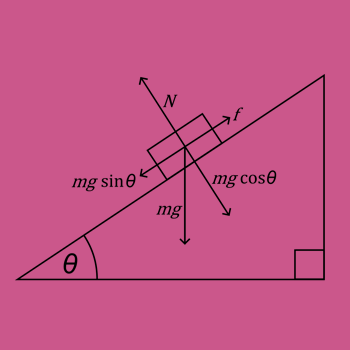
Question #fc5c1
1 Answer
Explanation:
Given:
The volume and concentration of a substance are related to the number of moles by the equation
where
The number of moles of a substance is related to its mass by the equation
where
Find the number of moles of solution using the first equation:
Find the mass using the second equation: