Can the ground state energy of an electron be negative?
1 Answer
Ground state energy not only can be negative, it is negative.
Explanation:
The energy of an electron is zero when the electron has completely left the atom, i.e., when its principal quantum number n = ∞.
When the electron is bound to the atom with any closer value of n, its energy is lower and is therefore negative. Thus, all energy states of an electron, including the ground state, have negative energies.
The ionization energy — the energy required to remove an electron from an atom and raise it to 0 J — of an H atom is 13.60 eV or 2.179× 10⁻¹⁸ J. Therefore, the ground state energy of an electron in an H atom is -13.6 eV or -2.179 × 10⁻¹⁸ J.
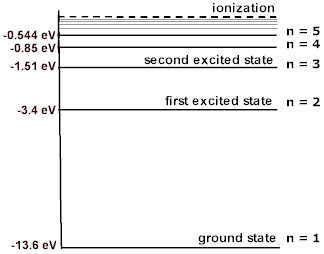
Please note that the reason the energy value is negative is related to the way the potential energy of the electron is defined:
#V(r) = (q_1q_2)/(4piepsilon_0r)#
#epsilon_0# is a positive constant,#8.854 xx 10^(-12) "F"cdot"m"^(-1)# .#r# is a positive radial distance in#"m"# .#e# is the charge of the proton (#1.602 xx 10^(-19) "C"# ), and since the electron interacts with the positive nucleus, one charge#q_1=e# is positive and the other,#q_2=-e# , is negative, leaving a negative sign in front of#V# .
All remaining quantities are positive, so here,
For further reading, see What is potential energy?.


