Question #a36d7
1 Answer
The key to understanding the formation of the ammonium ion (
In a simple covalent bond, two atoms share a pair of electrons. Each of these two atoms contributes an electron to the bond, the way the resulting bonding electron pair is shared being determined by the atoms' electronegativities.
However, there are cases when one atom supplies both electrons to the bond, while the other one supplies none. This is called a dative covalent bond, or a co-ordinate covalent bond. This is what happens in the case of
Let's analyze the reaction between ammonia (
The reaction between ammonia and hydrochloric acid takes place in aqueous solution; since
Here comes the interesting part.
The lone pair on nitrogen will be attracted to the
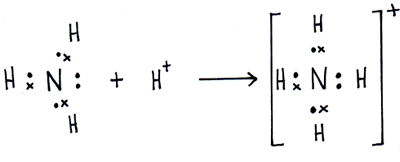
The ammonium ion is formed by the protonation (the addition of a proton) of ammonia. Therefore, the ammonium ion does not throw away an electron, since it did not bond with a fourth hydrogen atom, it bonded with a proton,
Here's a video on dative covalent bonds and the formation of the ammonium ion:

