Question #fa36e
1 Answer
The density of a substance is its mass per unit of volume.
The density formula is:
To solve for volume, multiply both sides of the equation times volume. This will cancel volume on the right and place it on the left.
Now, divide both sides by the density.
Density cancels on the left side, so that
[Mass units are typically grams (
There is a device called a density triangle that can make it easier to determine how to solve for one variable when the other two are known.
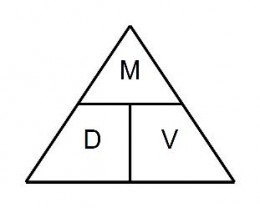
M = mass, D = density, and V = volume. If you want to find density when mass and volume are known, cover the D, leaving the M over the V, which means to divide the mass by the volume to get the density. If you want to find mass when density and volume are known, cover the M, leaving the D and V next to each other. That means to multiply density times volume to get mass. If you want to find volume when mass and density are known, cover the V, leaving the M over the D, which means to divide the mass by the density to get volume.

