Question #506f6
1 Answer
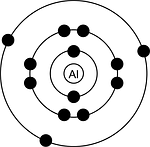
There are 3 valence electrons.
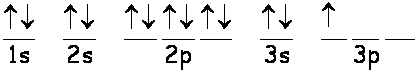
The electronic structure of aluminium is:
There are 3 electrons in the outer n = 3 level and so these are the valence electrons.
A commenter has asked about the tetrachloroaluminate ion. It can be regarded as having this structure:

For VSEPR purposes there are the 3 valence electrons from aluminium, 3 from 3 of the chlorines and 2 from a
A mechanism by which it forms occurs when you add aluminum in
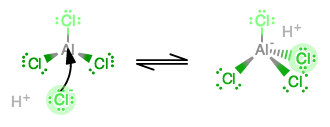
At first aluminum radically donates one electron and chlorine donates one. That happens three times, and then the fourth chlorine has to donate two electrons instead of just one in order to complete aluminum's octet.
At this point, the molecular geometry changes from trigonal planar to tetrahedral.


