Question #d6441
1 Answer
A limiting reagent, like its name also suggests, is a compound that limits a reaction, i.e. limits how much products can be formed.
In every chemical reaction that involves more than one reactant, the mole ratios that exist between all the species involved are constant at all times, regardless of the amount of reactants used.
Here's a generic reaction as an example
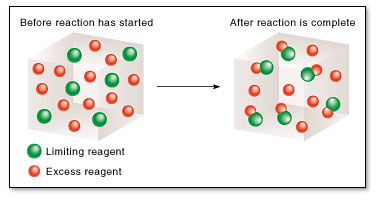
Notice that you have two kinds of reactants in the first cube - green ones and red ones. The reaction will pair one green ball with one red ball - this is the equivalent of having a
After the reaction takes place, 6 red-green pairs are formed and 6 red balls are left unpaired. This happens because the green balls acted as a limting reagent - they determined exactly how many red balls will be consumed and, consequently, how many red-green pairs will be produced.
This is what limiting reagent basically means - an insufficient amount of a reactant when compared with the minimum quantity established by the mole ratio.
The above reaction can be looked at from two perspectives - you can say you have insufficient green balls - limiting reagent, or you can say you have excess red balls - excess reactant.
According to the
If, for example, you would have had 20 green balls and 12 red balls, the red balls would have been the limiting reagent, since now only 12 of the 20 green balls available reacted
Here's a nice analogy made with burgers

The buns, slices of cheese, leaves of lettuce, and 5 pieces of burger meat are all reacting to produce burgers. Assuming you use one of each to make a burger, the limiting reagent will be the lettuce leaves.
The "reaction" will produce 3 burgers - the amount determined by the limiting reagent - and have 1 bun, 3 slices of cheese, and 2 pieces of meat in excess.

