Question #46b0b
2 Answers
Two of the dichlorocyclobutane isomers are optically active: (
Explanation:
Let's look at the possibilities.
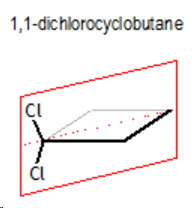
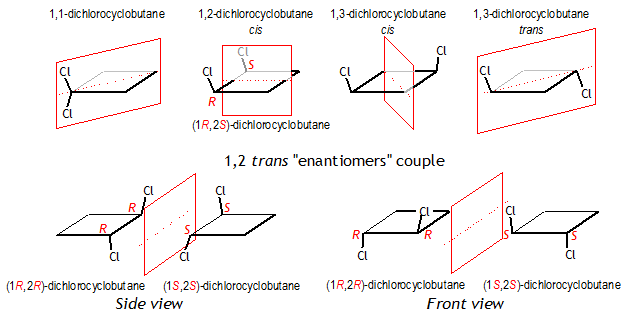
1,1-Dichlorocyclobutane
(From www.chem.yale.edu)
This does not have a chiral carbon, so it is not optically active.
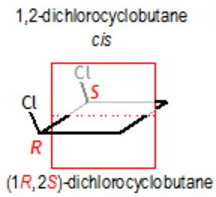
cis-1,2-Dichlorocyclobutane
(From www.chem.yale.edu)
This has two chiral carbons, but it has an internal plane of symmetry that bisects the C1-C2 and C3-C4 bonds.
It is a meso compound, so it is not optically inactive.
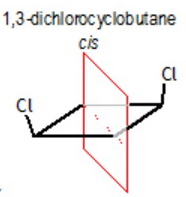
cis-1,3-Dichlorocyclobutane
(From www.chem.yale.edu)
This has no chiral carbons, so it is not optically active.
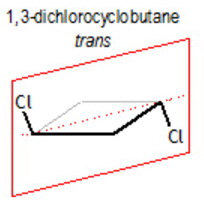
trans-1,3-dichlorocyclobutane
(From www.chem.yale.edu)
This has no chiral carbons, so it is not optically active.
trans-1,2-Dichlorocyclobutane
(From www.chem.yale.edu)
trans-1,2-Dichlorocyclobutane has two chiral carbon atoms, and it lacks an internal plane of symmetry.
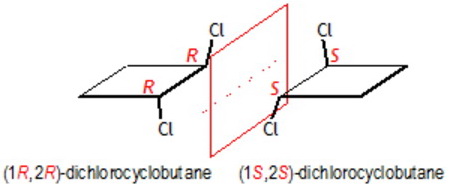
It is not superimposable on its mirror image, so it exists at two enantiomers: (
Thus, only two of the isomeric dichlorocyclobutanes are optically active.
There are six dichlorocyclobutanes, two of them are optically active.
The two optically active molecules are the two trans-1,2-dichlorocyclobutanes. They have opposite optical activity (ability to rotate plane-polarized light, clockwise = (+) or counterclockwise, (-)) and they are a couple of enantiomers (two molecules that are identical in every respect, as they are mirror images of each other, but not-superposable or not-identical; i.e. they are like the left and right hands).
As you can see from the picture below, the four isomers that are not optically active, are symmetrical or " achiral " molecules (they have symmetry planes and rotational axes giving exactly the same molecule after rotation or reflection). The two enantiomers are "chiral", or asymmetrical molecules and this causes their opposite ability to interact with [polarized light] beams. (http://en.wikipedia.org/wiki/Chirality_%28chemistry%29)
To distinguish one trans 1,2dichlorocyclobutane from the other, we can use IUPAC nomenclature, which have adopted the Cahn - Ingold - Prelog rules , or CIP system , in which every carbon having four different substituents is a stereogenic carbon (as there are in 1,2-dichlorocyclopropanes only) the carbon atom can be " R" or "S".
The rigidity of the four tetrahedral bonds prevent such carbon atom to pass from "R" to "S" or vice versa.
There is no way to deduce which enantiomer will be "levorotatory" (-) and which "destrorotatory" (+) from R or S nomenclature. This is an experimental matter.
The cis 1,2-dichlorocyclobutane has two of such stereogenic carbons, but is achiral, because of its internal symmetry. For this reason it has not another enantiomer (it is superposable or identical with its mirror structure), and is called a " mesoform".


