Is 1,2-dimethylcyclohexane a meso compound?
1 Answer
Jun 2, 2015
Only the cis isomer of 1,2-dimethylcyclohexane is a meso compound.
A meso compound must have at least two chiral centres but still be optically inactive because of an internal plane of symmetry.
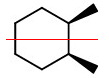
We see that the cis isomer has chiral carbons at
It also has a plane of symmetry passing through the mid-points of the 1,2 and 4,5 bonds
So the cis isomer is a meso compound.
In the trans isomer, one of the bonds is dashed.
The mirror plane vanishes, so the trans isomer exists as a pair of enantiomers.

