Question #f0437
1 Answer
Apr 29, 2015
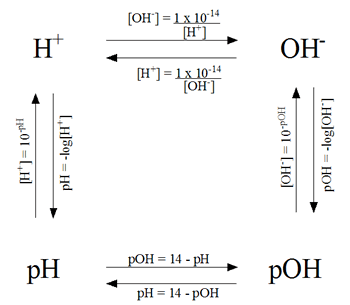
In one word: yes.
If you know the concentration of the hydroxide ions, you can determine the solution's pOH by
This will indirectly get you the pH of the solution
Now use the pH to determine the concentration of the hydronium ions
Alternatively, you can calculate the concentration of the hydronium ions directly by
Here's a diagram that shows how pH, pOH,