Question #2d154
1 Answer
In order of increasing radius, you'll have
The general trend for atomic size is that atomic radius decreases as you move from left to right across a period of the periodic table.
This happens because the attraction between the nucleus and the electrons that surround it increases as you move from left to right across a period - this is referred to as effective nuclaer charge.
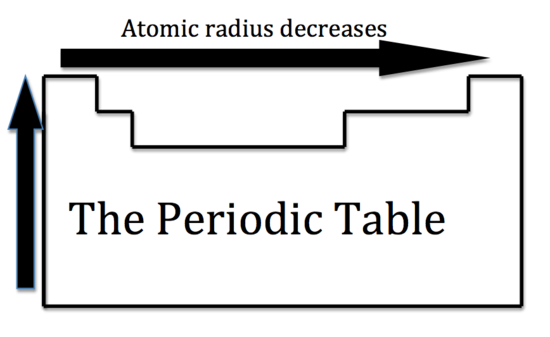 http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Descriptive_Chemistry/Periodic_Trends_of_Elemental_Properties/Periodic_Properties_of_the_Elements
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Descriptive_Chemistry/Periodic_Trends_of_Elemental_Properties/Periodic_Properties_of_the_Elements
Since lithium is located in group 1, it will have the largest atomic radius, even when compared with the oxygen anion,
Since an anion is always bigger than its parent atom, and since fluorine comes after oxygen in the period, the fluorine atom will be the smallest of the three.
The correct order will be

