Question #77567
1 Answer
The molecules you listed don't match that description. Here's why.
Right from the get-go, ozone, ammonia and water are eliminated because they are polar molecules. This leaves you with nitrogen,
Nitrogen is a homonuclear diatomic molecule formed when two nitrogen atoms form a covalent bond.
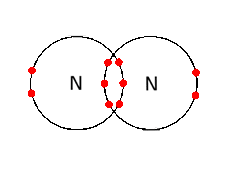
Since the bond is formed between two identical nitrogen atoms, the bonding electrons will not be more attracted to one of the two atoms,. This means that the bond will be nonpolar covalent, i.e. the bonding electrons will be shared equally between the two atoms.
Methane's Lewis structure looks like this
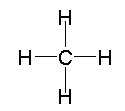
Now, a difference in electronegativity between carbon and hydrogen does exist, but it's too small for the
As a rule of thumb, if the difference in electronegativity between two atoms is smaller than 0.5, the bond is said to be nonpolar.
So, if you were to consider the

