Question #0e8fa
1 Answer
To get an idea on what type of bond two atoms form when bonded together, you can use the two atoms' electronegativity values, or, more specifically, the difference in electronegativity values.
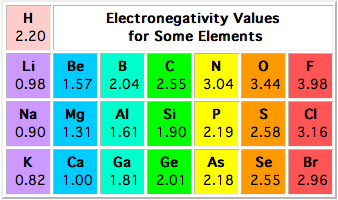
According to the Pauling Electronegativity Scale, hydrogen has an electronegativity of 2.20 and phosphorus has an electronegativity of 1.19.
The difference in electronegativity between the two atoms will be
Because this difference is so small, the bond between the two atoms will essentially be nonpolar covalent. For all intended purposes, the two bonding electrons that make up the
You can confirm this by calculating the %ionic character of the bond
In your case, the ionic character of the
This means that your bond is 99.9975% nonpolar covalent.

