Question #f848e
1 Answer
In order of increasing bond polarity :
Explanation:
Bond polarity is all about unequal sharing of the bonding electrons.
When two atoms that are bonded together don't share their respective bonding electrons equally, then a separation of charge forms.
If the bonding electrons spend more time around one of the two atoms, then that atom will develop a partial negative charge. Likewise, the other atom will develop a partial positive charge.
Now, in order to predict which of the two atoms, if any, will attract these bonding electrons more you have to look at electronegativity values.
An atom's electronegativity essentially tells you how capable of attracting bonding electrons that respective atom will be.
So, in your case, you have three different atoms, carbon, phosphorus, and chlorine, all bonded to oxygen.
The periodic trends in electronegativity are as follows
- electronegativity increases when moving left to right across a period
- electronegativity decreases when moving top to bottom in a group
So, use these two trends to see if you can establish a relationship between the electronegativity values of these atoms.
Oxygen and carbon are in the same period, so the one further to the right, in this case oxygen, will be more electronegative.
The same goes for phosphorus and chlorine, whith chlorine, the one further to the right, being more electronegative.
You know that
None of your four atoms share a group, but you can use the fct that second-period atoms have larger atomic radii to determine that the electronegativity of phosphorus will be smaller than that of carbon and the electronegativity of chlorine will be smaller than that of oxygen.
So, if you combine all this information, you'll get
Now for the polarity of bonds. The bigger the difference between oxygen and the other atom's electronegativity values, the more polar the bond will be.
This means that, in order of increasing polarity, you can arrange these bonds like this
Alternatively, you can consult the actual electronegativity values
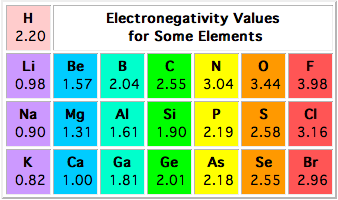
and confirm that we've got the order right.

