Question #f5310
1 Answer
Explanation:
The ammonium ion is formed when a molecule of ammonia,
This tells you two things
- the ammonium ion will have a total of four hydrogen atoms, one more than the ammonia molecule
- the ammonium ion will have an overall
#(1+)# net charge coming from the added proton*
This means that the chemical formula for the ammonium ion will be
#"NH"_4^(+)#
The hydronium ion is formed when a molecule of water,
This of course means that the hydronium ion will have three hydrogen atoms and a net overall charge of
#"H"_3"O"^(+)#
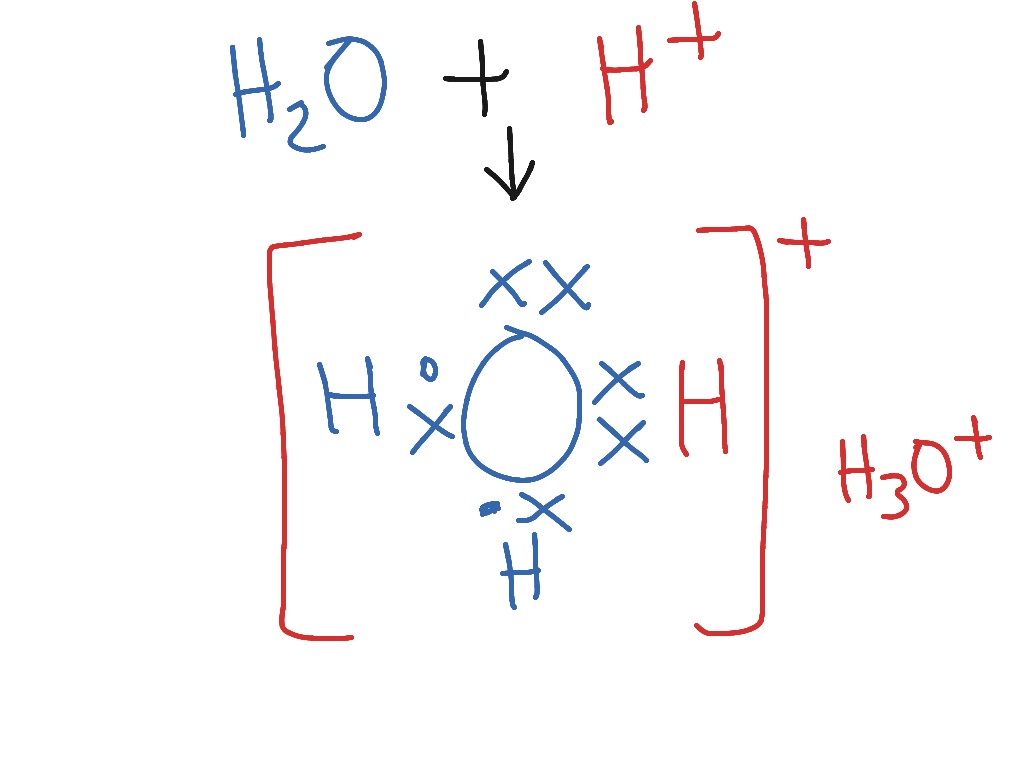
An alternative way of thinking about these ions is that they represent the conjugate acids of their respective bases, ammonia and water.
When a base accepts a proton from an acid, the compound that's formed is called a conjugate acid. In this case, the ammonium ion is the conjugate acid of ammonia and the hydronium ion is the conjugate acid of water.

