Question #da38a
1 Answer
Jan 3, 2016
The conjugate bases are
Explanation:
According to the Brønsted-Lowry theory, an acid
Its conjugate base
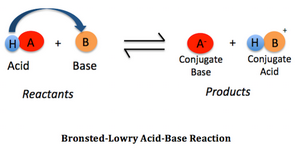
(from study.com)
The equation for
The conjugate base is
The equation for
The conjugate base is

