Here is a short #pK_"a"# table.
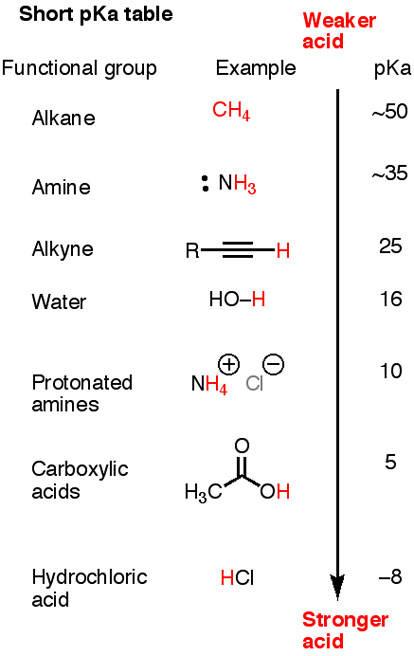
Note that #"HC≡C-H", "H"_2"N-H"#, and #"H"_3"C-H"# are all weaker acids than water.
The corresponding conjugate bases, #"H-C≡C:"^"-", "H"_2"N:"^"-"#, and #"H"_3"C:"^"-"#, are strong. They will all deprotonate water.
The conjugate base of methane #"H"_3"C:"^"-"# is strong enough to deprotonate anything below it in the table.
Methyllithium, #"CH"_3"Li"#, is one of the strongest bases around.
Acetylide ion (#"H-C≡C:"^"-"#) is strong enough to deprotonate only an acid with a #"pK"_"a" < 25#.
Acetate ion (#"CH"_3"COO"^"-"#) is weaker still, able to deprotonate only an acid with a #pK_a < 5#.
A more complete table of #pK_"a"# values will give you many more conjugate bases that will deprotonate water.


