Question #6f5cf
1 Answer
Explanation:
So, in order to write the structure of this coordination complex, you need to be familiar with the nomenclature rules used to name complex ions.
Let's break it down step by step to see if we can figure out what this coordination complex looks like. SO, you know that the cation is the bromide anion,
Now, notice that the name of the complex cation includes a Roman numeral. As you know, Roman numerals are used in naming ionic compounds to signal the oxidation state of the transition metal cation.
The exact same principle applies here as well. In your case, the transition metal cation will be nickel(II),
Now, aqua is the name given to water when it acts as a ligand. As you know, Greek prefixes are used to signal multiple instances of the same ligand.
This means that your complex ion will feature a
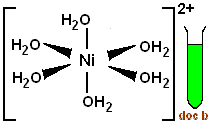
The coordination sphere, which consists of the metal cation and its attached ligands, will thus look like this
#["Ni"("H"_2"O")_6]^(2+)#
Another important thing to notice here is that water molecules do not carry a net charge, i.e. they are neutral. This means that the charge of the complex ion will be equal to that of the nickel cation,
As a result, you will need two bromide anions to balance the charge of the complex ion, since the bromide anion carries a
#["Ni"("H"_2"O")_6]"Br"_2#

