Question #3888e
1 Answer
Here's what I got.
Explanation:
Before going into crystal field theory and ligand strength fields, it's worth taking a second to understand what paramagnetic and diamagnetic mean.
Basically, paramagnetic and diamagnetic refer to the way a chemical species interacts with a magnetic field. More specifically, it refers to whether or not a chemical species has any unpaired electrons or not.
A diamagnetic species has no unpaired electrons, while a paramagnetic species has one or more unpaired electrons.
Now, I won't go into too much detail about crystal field theory in general, since I assume that you're familiar with it.
So, you're dealing with the hexafluorocobaltate(III) ion,
You know that
Both complex ions contain the cobalt(III) cation,
#"Co"^(3+): 1s^2 2s^2 2p^6 3s^2 3p^6 color(blue)(3d^6)#
For an isolated cobalt(III) cation, all these five 3d-orbitals are degenerate. The thing to remember now is that the position of the ligand on the spectrochemical series will determine how these d-orbtals will split.
More specifically, you can say that
- a strong field ligand will produce a more significant splitting energy,
#Delta# - a weak field ligand will produce a less significant splitting energy,
#Delta#
Now, the spectrochemical series looks like this
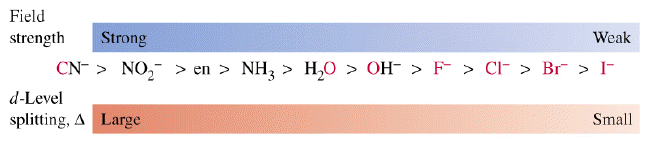
Notice that the cyanide ion,
In the case of the hexafluorocobaltate(III) ion, the splitting energy is smaller than the electron pairing energy, and so it is energetically favorable to promote two electrons from the
This will ensure that the hexafluorocobaltate(III) ion will have unpaired electrons, and thus be paramagnetic.
On the other hand, in the case of the hexacyanocobaltate(III) ion, the splitting energy is higher than the electron pairing energy, and so it is energetically favorable to pair up those four electrons in the
Since it has no unpaired electrons, the hexacyanocobaltate(III) ion will be diamagnetic.

