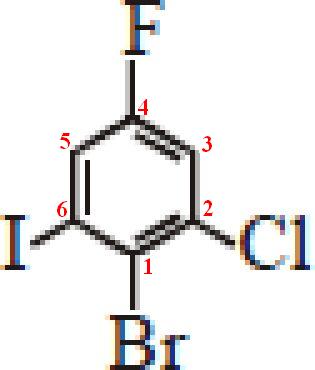
Question #861f9
2 Answers
1-bromo-2-chloro-4-fluoro-6iodobenzene
Explanation:
When naming an aromatic compound that has more than one substituent, you need to assign relative positions around the ring for these substituents.
Also, it is crucial to remember that substituents are always listed in alphabetical order in the name of the compound. In your case, arranging these four substitutes in alphabetical order would get you
#color(blue)("b")"romo" -> color(blue)("c")"hloro" -> color(blue)("f")"luoro" -> color(blue)("i")"odo"#
Now, the lowest number is given to the first substituent. In your case, bromo would get
The second substituent in alphabetical order, chloro, would get
At this point, you can start to number the positions in an anticlockwise direction.
The third substituent, fluoro, would get
Finally, Iodo would get
Remember, we all labeling these groups in anticlockwise direction.
Finally, the name of the compound will be formed using these positions and the substituents listed in alphabetical order.
1-bromo-2-chloro-4-fluoro-6-iodobenzene
The IUPAC name is Option 2: 2-bromo-1-chloro-5-fluoro-3-iodobenzene.
Explanation:
None of the substituents takes priority over the others, so the only problem is in numbering the ring.
Where do we start?
The rule is to choose the lowest possible numbers as we go around the ring.
That gives us the sequence 1, 2, 3, 5.
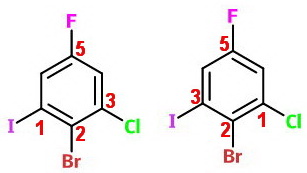
However, there are two possible sequences. Do we go clockwise or counterclockwise?
The choice is 1-iodo or 1-chloro.
Here, the alphabetical sequence takes precedence, and we choose 1-chloro.
However, the substituents are listed in alphabetical order in the name.
Hence, the IUPAC name is 2-bromo-1-chloro-5-fluoro-3-iodobenzene.